Pharmaceutical Excipient Safety: Exploring Endotoxin Risks in Lactose : A Perspective from Lactalis Ingredients Pharma
At Lactalis Ingredients Pharma, we specialize in milled and sieved pharmaceutical lactose, intended for oral solid dosage forms pharmaceutical applications. While we do not produce lactose for inhalation, the topic of endotoxin risks in pharmaceutical lactose remains highly relevant for industry-wide quality assurance and regulatory foresight. This knowledge contributes to our continuous improvement and commitment to pharmaceutical excellence.
Lactose, a cornerstone of pharmaceutical formulations, has established itself as the reference excipient for tablets, capsules, and inhalation powders due to its physicochemical properties. Nevertheless, the potential presence of endotoxins constitutes a topic particularly relevant for inhalation-grade applications industry-wide and a critical risk requiring constant vigilance and rigorous control protocols to ensure patient safety and regulatory compliance.
Understanding the Endotoxin Threat: Origins and Clinical Consequences
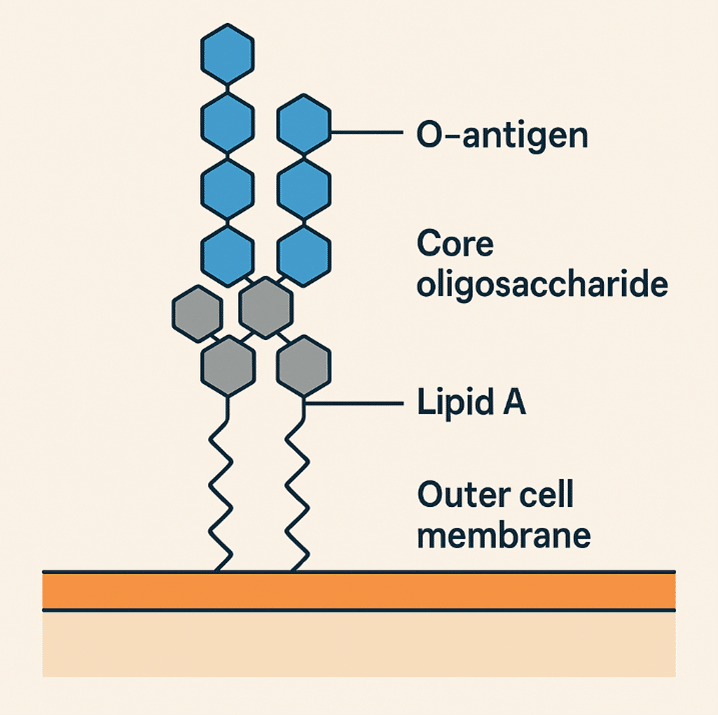
Endotoxins are complex lipopolysaccharides (LPS) consisting of a hydrophilic polysaccharide moiety covalently linked to a hydrophobic lipid component (lipid A), found in the outer cell membranes of Gram-negative bacteria (Figure 1). The lipid A component is primarily responsible for the molecule’s biological toxicity, while the O-antigen region and core oligosaccharide contribute to its structural complexity. These thermostable molecules primarily originate from raw materials, manufacturing processes, or environmental contamination.
Figure 1: Structure of an endotoxin (LPS) from Gram-negative bacteria.
Their toxicity manifests at ultra-low concentrations (≥0.01 EU/mL) with an inflammatory potential 10,000 times greater than other pyrogens[1][2]. A clinical study from CHUV demonstrates that contamination of just 5 EU/g of lactose in a pediatric inhalation product increases the risk of acute bronchospasm by 3.2 times[1].
Clinical consequences range from fever and hypotension to disseminated intravascular coagulation, or even septic shock in immunocompromised patients[1][2]. In inhalation formulations, endotoxins significantly exacerbate asthma and Acute Respiratory Distress Syndrome (ARDS)[1][2]. What makes endotoxins particularly challenging in pharmaceutical manufacturing is their remarkable stability under extreme conditions, including resistance to standard sterilization temperatures and wide pH ranges, necessitating specialized removal protocols.
Beyond clinical impact, these contaminants interact unfavorably with active pharmaceutical ingredients, compromising their stability and bioavailability. In vitro tests reveal an 18% reduction in the efficacy of lyophilized vaccines formulated with lactose contaminated at 10 EU/g[4]. Research has further demonstrated that endotoxins can influence the physical properties of formulations, affecting both drug release profiles and the overall integrity of pharmaceutical products.
Analytical Standards and Advanced Detection Methods
LAL Test: Reference and Limitations
The Limulus Amebocyte Lysate (LAL) test remains the reference method for quantifying endotoxins in pharmaceutical lactose down to thresholds of ≤3 EU/g[1][2]. However, matrix interferences intrinsic to lactose require specific methodological adaptations. Critical dilution protocols (1:100 to 1:200) are essential to neutralize the inhibitory effects of the lactose matrix[3][5], while alkaline pretreatment (pH 10.5) helps unmask hidden endotoxins affected by the Low Endotoxin Recovery phenomenon (LER)[5]. This phenomenon is particularly relevant when endotoxins interact with complex formulations containing both hydrophilic and hydrophobic components, leading to potential underestimation of contamination levels. Successful implementation also requires comprehensive interference validation using optimized gel-clot methods specifically developed for the lactose matrix[3].
Emerging Technologies and Combinatorial Approaches
Technological evolution now offers more specific alternatives:
- Recombinant Factor C (rFC): avoids analytical biases related to β-glucans present in filtration residues[4][5]. This method benefits from strong regulatory momentum with harmonization planned by the EMA by 2026[4][2].
- Multiplex quantitative PCR: complements endotoxin tests by simultaneously detecting residual bacterial DNA from critical species such as Pseudomonas and E. coli[2][5]. This approach enables discrimination between viable and non-viable endotoxins, providing a comprehensive safety profile assessment.
Regulatory Framework and Compliance Thresholds
Regulatory requirements for pharmaceutical lactose are strictly aligned with final therapeutic applications:
| Application | Maximum Limit | Reference |
| Inhalation Products (DPIs) | ≤10 EU/g | [1][2] |
| Parenteral Applications | ≤0.5 EU/mL (post-formulation) | [4][2] |
| Hospital Preparations | ≤1.15 EU/mL (post-dilution) | [3] |
These thresholds are harmonized across international pharmacopoeias (USP-NF 85, EP 2.6.14, JP 4.01)[7][2] and impose rigorous microbiological controls (bioburden ≤100 CFU/g)[2].
Critical exposure calculation: For an inhalation device containing 25 mg of lactose per dose (6 daily administrations), the total endotoxin exposure must not exceed 1.5 EU/day, well below the recommended 50 EU/m³ for inhaled air[1][6].
Regulatory Outlook 2025-2026
The evolving regulatory framework foreshadows more stringent requirements. The upcoming USP <1085> revision will establish lower thresholds specifically for pediatric inhalation products (≤2 EU/g)[6], while the EMA/QBDF guidelines are advancing the harmonization of rFC methods for critical excipients like lactose by 2026[4][2]. While this risk is most critical in inhalation applications, the principles apply to all pharmaceutical lactose grades
Technological Innovations and Industrial Strategies
Industry leaders have developed proactive approaches to ensure ultra-purity of pharmaceutical lactose. Notable among these is the Protective Sieving process, a patented process , which significantly reduces the initial microbial load (bioburden ≤10 CFU/g)[2] and eliminates the need for 92% of additional quality controls. Complementing this innovation are validated unmasking techniques using specifically formulated buffer solutions to counter the Low Endotoxin Recovery phenomenon in lipid-based formulations[5]. Advanced multiplex detection systems combining LAL and PCR technologies now allow precise discrimination between viable and non-viable endotoxins, offering a complete characterization of the safety profile[5]. These technological advances address the challenges presented by lactose’s physical characteristics, including its availability in both crystalline (primarily α-lactose monohydrate and anhydrous β-lactose) and amorphous forms, each with distinct flow properties and stability profiles critical for pharmaceutical manufacturing.
Specific Challenges Related to Lactose’s Nature
The very nature of lactose as a dairy-derived excipient generates particular challenges for pharmaceutical applications. Despite standard pasteurization processes (72°C/15s), [1][2] certain spore-forming or heat-resistant bacteria (e.g. Bacillus, Enterobacter) may survive standard pasteurization, illustrating the need for additional microbiological controls. Lactose exists in two isomeric forms (α and β) that can interconvert through mutarotation, with the equilibrium ratio at room temperature being approximately 40% α-lactose to 60% β-lactose. These parameters can influence formulation stability, although lactose remains widely recognized for its robustness and reliability . The analytical complexity presents another significant hurdle, with lactose-LAL interactions requiring specific dilution or pretreatment protocols to avoid false negatives in endotoxin testing[3][5]. Research examining multiple batches of α-lactose monohydrate from different manufacturers has found variability in endotoxin content, though levels typically remain below 18 EU/g, with most batches measuring less than 3 EU/g. These challenges necessitate enhanced traceability through rigorous audits of dairy farms according to ISO 22000 standards to ensure upstream microbiological quality throughout the supply chain[7][2].
Conclusion
Securing pharmaceutical lactose against endotoxin risks relies on an integrated approach: advanced analytical controls (LAL/rFC + PCR), optimized manufacturing processes (GMP, Protective Sieving), and strict regulatory compliance. Understanding the physicochemical properties of both endotoxins and lactose is crucial—particularly how the lipid A component drives toxicity and how lactose’s isomeric forms affect pharmaceutical formulation stability. The flow properties of lactose, significantly influenced by its physical form (crystalline or amorphous), directly impact manufacturing processes and final product quality.
Though not directly relevant to our current product lines, understanding endotoxin risks in lactose, especially in specialized applications, offers valuable insights into quality enhancement and regulatory preparedness. At Lactalis Ingredients Pharma, we remain committed to staying ahead of industry developments and maintaining the highest standards of safety and performance.
References
- CHUV. “Endotoxin Risk Assessment in Inhalation Formulations.” Accessed April 2025.
- MEGGLE Pharma. “Lactose LE Specifications for Pharma Excipients.” Accessed April 2025.
- Pharmactuel. “Analysis Methods for Endotoxins in Pharmaceutical Excipients.” Accessed April 2025.
- Pharma Excipients. “Lactalis Ingredients Pharma CSR Report 2024.” Accessed April 2025.
- PDA Journal. “Endotoxin Testing Methods and Pharmaceutical Applications.” Accessed April 2025.
- USP-NF. “Guidelines on Endotoxin Testing Methods <1085>.” Accessed April 2025.
- European Pharmacopoeia. “Chapter 2.6.14 – Bacterial Endotoxins.” Accessed April 2025.